 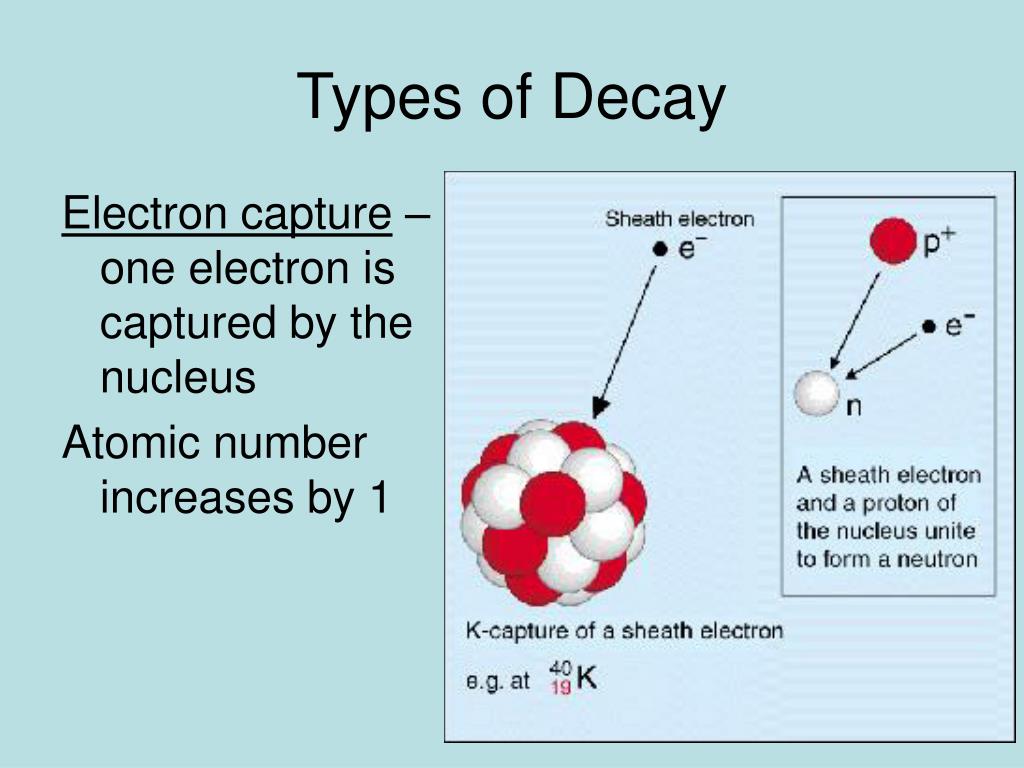
Proton-deficient nuclei undergo beta decay - emitting a beta particle (electron) and an antineutrino to convert a neutron to a proton - thus raising the elements atomic number Z by one. 67 Ga has a 3. Alpha decay occurs spontaneously only if the daughter and 4 He nucleus have less total mass than the parent. The daughter nucleus has two fewer protons and two fewer neutrons than the parent. Proton-deficient or neutron-deficient nuclei undergo nuclear decay reactions that serve to correct unbalanced neutron/proton ratios. 67 Ga has nuclear properties that make it potentially useful for diagnostic imaging applications. Figure 31.15 Alpha decay is the separation of a 4 He nucleus from the parent. Other heavy unstable elements undergo fission reactions in which they split into nuclei of about equal size. Write a balanced nuclear equation for the following The nuclide gallium-67 undergoes electron capture + - Submit Answer Retry Entire Group 1 more group attempt remaining When the nuclide iodine-123 decays by electron capture: The name of the product nuclide is The symbol for the product nuclide is 1 more group attempt remaining Retry Entire Grou. This article is focused on the calculation of electron-induced ionisation and total scattering cross sections by Boron, Aluminium and Gallium trihalide. Alpha decay is a form of spontaneous fission, a reaction in which a massive nuclei can lower its mass and atomic number by splitting. 
EC stands for electron capture, a ( annus, Latin) for years, h for hours, d for days, min. nator of this equation consists of the energy Em of the mth intermediate 1. Write the daughter nucleus product in the following nuclear processes: Beta emission of manganese-56 Gallium-67 decays by electron capture Potassium-38 decays by positron emission. Electron Capture: Electron capture is a type of nuclear decay where an electron ( 1 0 e) in the inner shell is drawn towards its. The energy released in an alpha decay reaction is mostly carried away by the lighter helium, with a small amount of energy manifesting itself in the recoil of the much heavier daughter nucleus. denotes the decay to the subsequent element in the ta- ble. 5Department of Physics, University of La Plata, C. Therefore, the mass of the parent atom must simply be greater than the sum of the masses of its daughter atom and the helium atom. The 0.092-Mev level of Zn 67 has a half-life of 9. Title: Microsoft Word - Gallium 67. Electron capture occurs to levels in Zn 67 at 0.092, 0.182, 0.39, and 0.87 Mev. Radiation: Decay mode: Electron Capture Major Betas: Major Gammas: Max E (MeV) Avg E (MeV) per 100 dis E (MeV) per 100 dis 0.093 36 None 0.185 20 0.300 16 Max. Write balanced nuclear equations for the following processes: (a) Polonium-207 undergoes positron decay. When electron capture occurs, the electron is a reactant. It is believed that the captured electron usually comes from the n 1 or n 2 levels. 
No positrons have been found, in agreement with the total decay energy of 1.003 Mev measured by Trail and Johnson. Electron capture is the capture of an electron by a nucleus. Since the number of total protons on each side of the reaction does not change, equal numbers of electrons are added to each side to make neutral atoms. The decay of Ga 67 has been investigated with scintillation spectrometers, single and in coincidence. Trending Questions What force did Wegner believe caused continental drift? Is a cherry blossom a gymnosperms? What can you use to reattach a toenail to the bed? What is the method of inquiry adopted by critical rationalists? A lipid bilayer is characteristic of all membranes? What are big bang member favorite color? Is 5 years for a volcanic eruption that could change Earth's surface is slow or a fast change? Does passive transport use a protein doorway? 29.\( \newcommand\]Īs with beta decay and electron capture, Δm must only be less than zero for spontaneous alpha decay to occur. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
